Hypertension Therapies and Renal Denervation
Long-Term Safety and Efficacy of Renal Denervation in the Global Symplicity Registry Using the Symplicity Spyral Catheter
Tzung-Dau Wang1, Markus Schlaich2, Felix Mahfoud3, Bryan Wiliams4, Luis Ruilope5, Krzystzof Narkiewicz6, Martin Fahy7, Giuseppe Mancia8, Michael Böhm3
National Taiwan University Hospital, Taiwan1, University of Western Australia, Australia2, Saarland University Hospital, Germany3, UCL, United Kingdom4, University Hospital October 12/ CIBERCV, Spain5, Medical University of Gdansk, Poland6, Medtronic, USA7, University of Milano-Bicocca and Policlinico di Monza, Italy8
The catheter-based renal denervation (RDN) procedure targetsoveractivity of the sympathetic nervous system to treat uncontrolled hypertension.Results from multiple recent randomized sham-controlled clinical trials havedemonstrated excellent safety and efficacy of RDN. We examined the long-term safetyand durability of the procedure in real-word patients.
The Global SYMPLICITY Registry (GSR) is a prospective,international registry of patients who received radiofrequency RDN treatment,primarily due to uncontrolled hypertension. Office and ambulatory bloodpressure (BP) levels were measured at baseline, 3, 6, 12, 24, and 36 months perstandard of care. Adverse events were collected out to 3 years.
Amongthe 641 patients treated with the Symplicity Spyral catheter as of March 2021,key demographic features include: baseline office systolic BP 168 ± 25 mmHg,4.6 ± 1.5 prescribed anti-hypertensive medication classes, meanage 60.5 ± 12.5years, 56.9% male, 42.5% history of cardiac disease, 37.2% typeII diabetesmellitus, and 19.1% renal insufficiency with eGFR<60ml/min/1.73m
2. At 3 years, there were no cases of new renal artery stenosis > 70%or renal artery re-intervention. Rates of other adverse events at 3 yearsincluded new-onset end-stage renal disease (2.4%), cardiovascular death (1.6%)and myocardial infarction (0.8%). Mean change in eGFR from baseline to 3 yearswas -6.5 ± 15.7 mL/min/1.73m
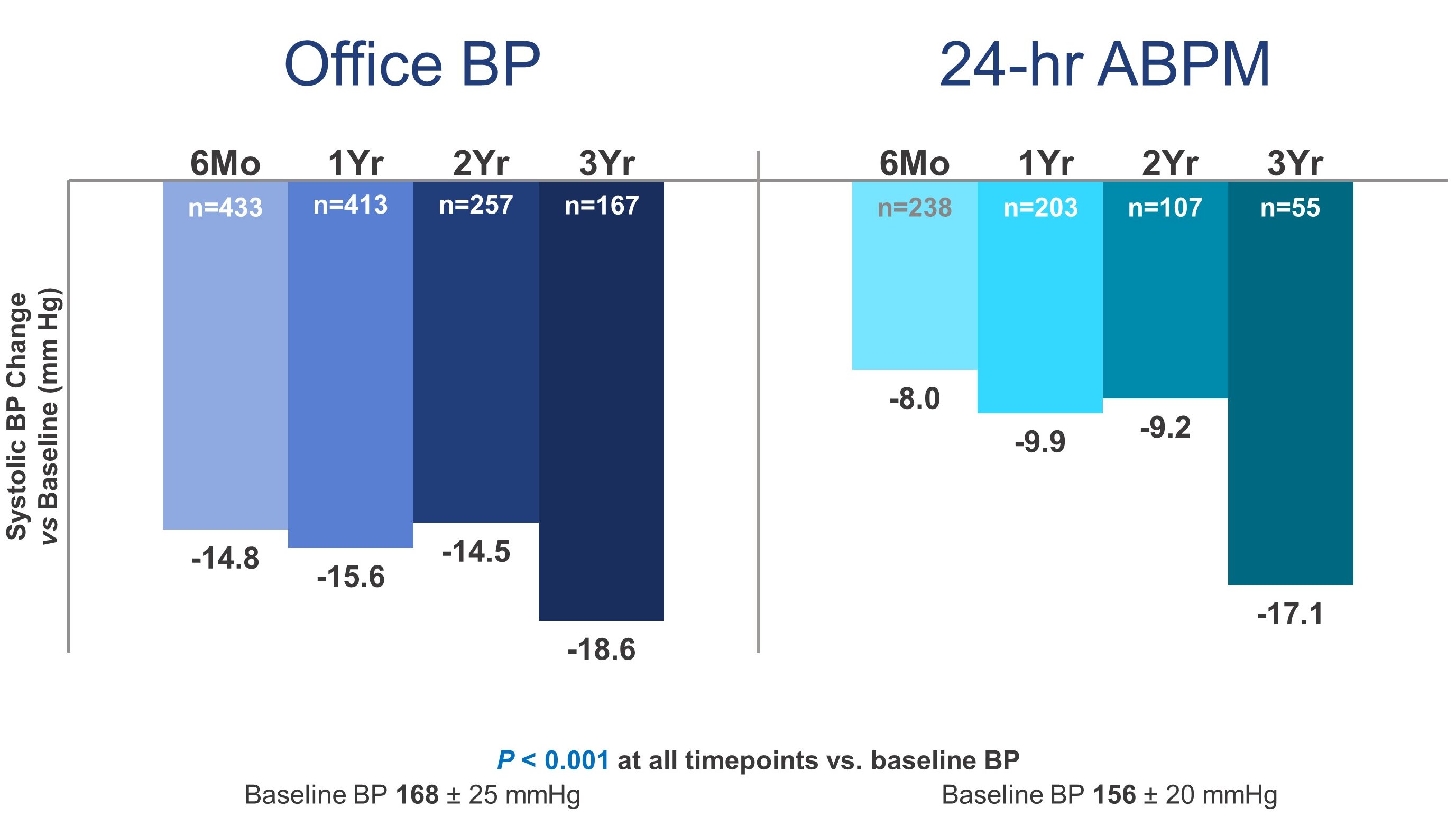
2. Significant reductions in mean 24-hour and officesystolic BP from baseline to 6, 12, 24 and 36 months are shown in the
Figure.Among patients with available follow-up at 36 months, the mean change in24-hour systolic BP from baseline was -17.1 mmHg (n=55) while the mean changein office systolic BP from baseline was -18.6mmHg (n=167). Diastolic office and24-hour BP also decreased significantly at 3 years, as did the number ofprescribed antihypertensive medications. Medications decreased from 4.75at baseline to 4.51 at 36 months (p=0.007).
Office and 24-hour BP weresignificantly reduced from baseline at all follow up time-points after RDN withthe Symplicity Spyral catheter, with no instances of renal arteryre-intervention.



