Lots of interesting abstracts and cases were submitted for TCTAP 2023. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge and interact with authors as well as virtual participants by sharing your opinion in the comment section!
TCTAP A-105
A New Method of Prosthesis Virtual Implantation for Pre-Procedural Assessment of TAVR: Comparison of Self-Expandable Devices in a Patient With Bicuspid Aortic Valve With Severe Stenosis and Calcification
By Xinlei Wu, Patrick W. Serruys, Tianbo Wu, Zhisheng Ruan, Weihao Xue, Daozhu Wu, Lianpin Wu, Xinmin Zhang
Presenter
Xinlei Wu
Authors
Xinlei Wu1, Patrick W. Serruys2, Tianbo Wu3, Zhisheng Ruan3, Weihao Xue3, Daozhu Wu1, Lianpin Wu1, Xinmin Zhang1
Affiliation
The Second Affiliated Hospital of Wenzhou Medical University, China1, University of Galway, Ireland2, Second Affiliated Hospital Wenzhou Medical University, China3
View Study Report
TCTAP A-105
Valvular Intervention: Aortic
A New Method of Prosthesis Virtual Implantation for Pre-Procedural Assessment of TAVR: Comparison of Self-Expandable Devices in a Patient With Bicuspid Aortic Valve With Severe Stenosis and Calcification
Xinlei Wu1, Patrick W. Serruys2, Tianbo Wu3, Zhisheng Ruan3, Weihao Xue3, Daozhu Wu1, Lianpin Wu1, Xinmin Zhang1
The Second Affiliated Hospital of Wenzhou Medical University, China1, University of Galway, Ireland2, Second Affiliated Hospital Wenzhou Medical University, China3
Background
Bicuspid aortic valve (BAV) patients with excess leaflet calcification had significantly higher mortality after transcatheter aortic valve replacement (TAVR) due to the higher rates of procedural complications and associated poorer outcomes. Patient-specific in silico simulation of TAVR based on computerized tomography images can predict paravalvular leakage after virtual implantation in order to improve the procedure outcomes. The aim of this study is to explore the feasibility of our new method of the prostheses virtual implantation for TAVR and compare treatment outcomes among several Chinese-made self-expanding prostheses virtually treated in a high-risk patient with type 0 BAV with severe stenosis and calcification.
Methods
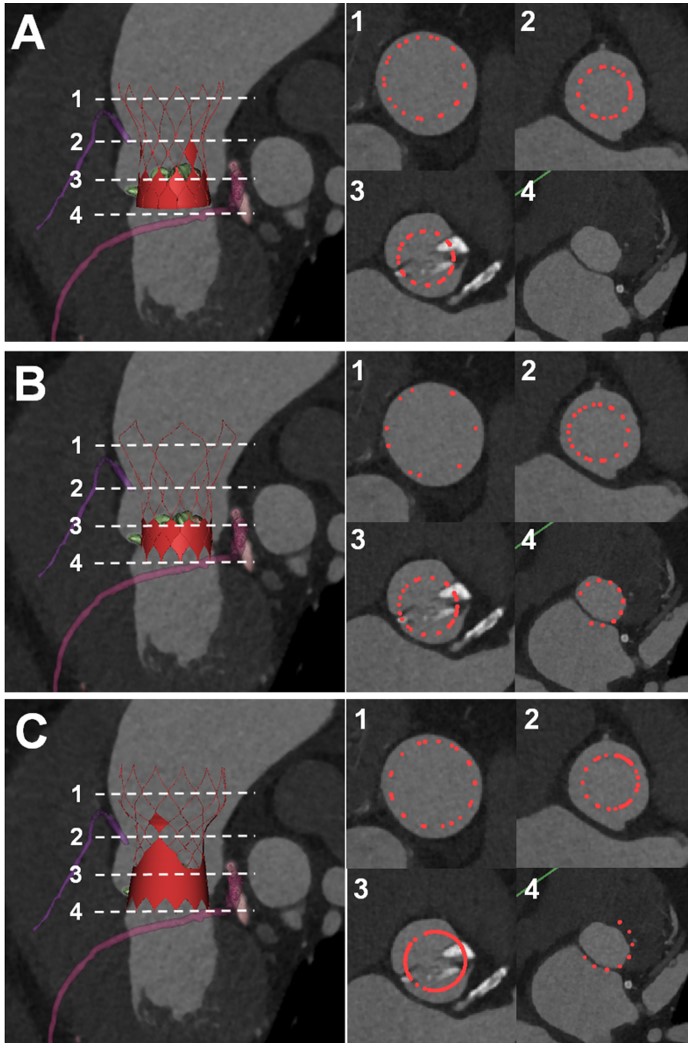
A series of in-silico models of self-expandable valves from the current three commercial companies (Peijia, Venus, and MicroPort) was constructed by a parameterized modeling method. The appropriate size of three prosthesis models was selected according to the quantitative analysis of the patient with BAV anatomy based on preoperative CT images and the “downsizing” principle due to the heavy calcification. To assess the potential risk of paravalvular leakage, anchoring effect, and coronary arterial occlusion of the individual treatment, the depths of three devices were virtually implanted at the standard position of the 3D reconstruction model of BAV and then quantitatively analyzed.
Results
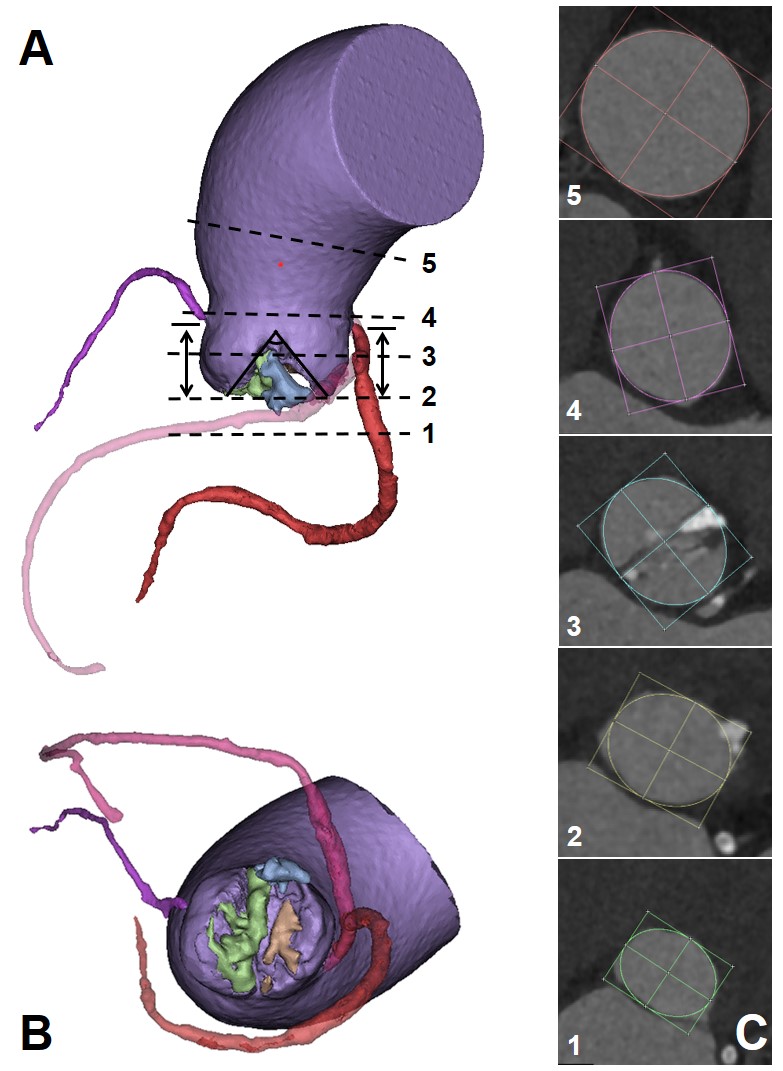
The aortic valve orifice area was only 0.46 cm2 with a severe calcification volume of 1828 mm3 (Figure 1). The right sinus had severe calcification and was unilaterally connected with the LVOT. The pronounced oval-shaped annulus of the BAV was observed with an average diameter (min: 23.2mm, max: 27.4mm, perimeter derived: 25.3mm). The ascending aorta is dilated with a diameter of 46.3mm at 40.0mm above the annulus. The right sinus is slightly larger than the left sinus, and the right coronary ostium (17.1mm) is higher than the left one (15.7mm). The heights of both ostia are higher than the curve length of the leaflets. The Venus model with the higher skirt and larger diameter of the annulus could have better sealing and anchoring effects at the two sinuses commissure and annulus, respectively (Figure 2). However, for all three models, the severe enlargement of the ascending aorta resulted in the malapposition of the upper parts of the prostheses, whereas the SOV has sufficient space and lowers the risk of coronary ostium occlusion (Figure 3).
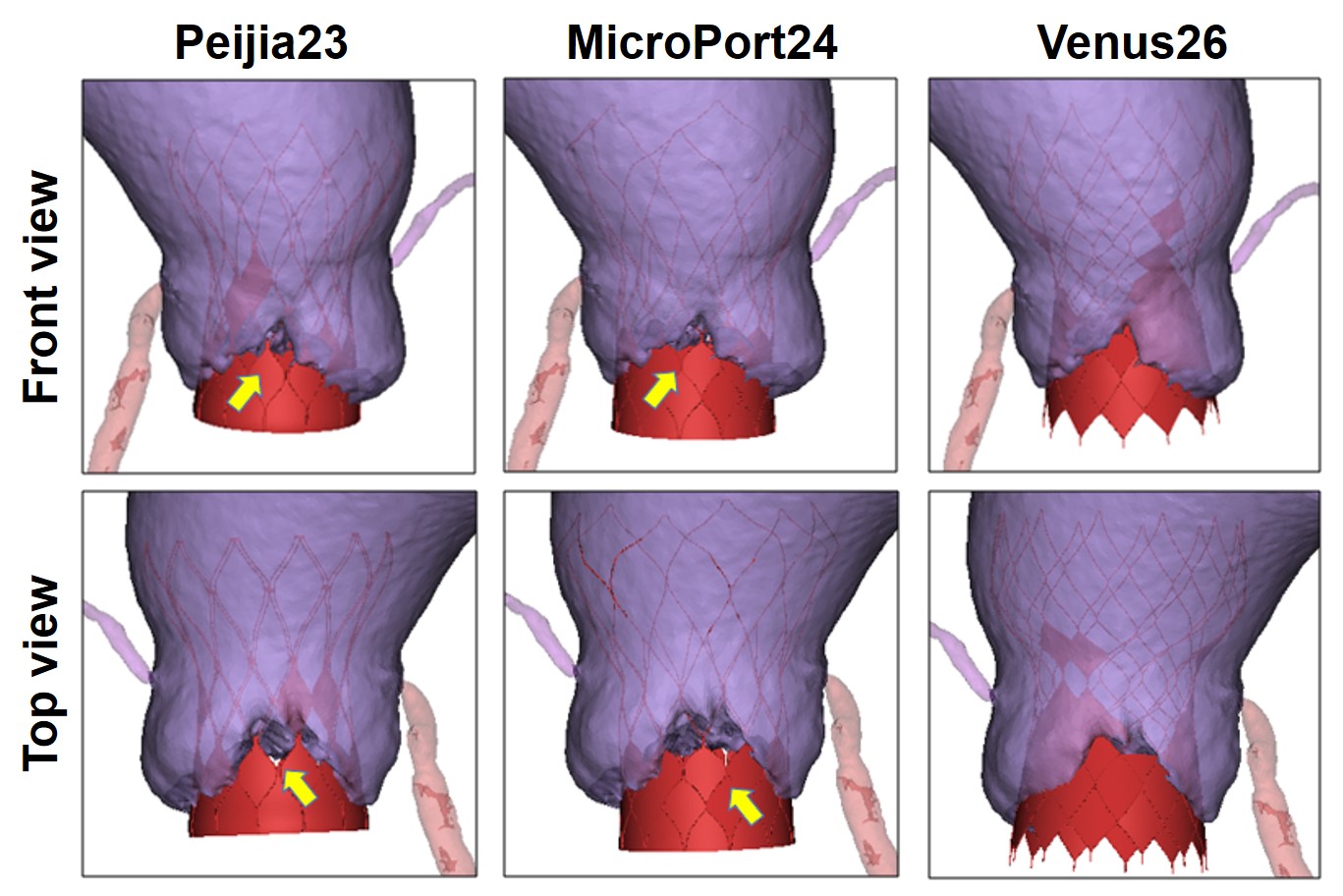



Conclusion
Compared with Peijia23 and MicroPort24, the Venus26 model with a larger diameter and higher skirt at the annulus level could be more suitable for this high-risk BAV patient when implanted at a standard position. Our new method of virtual implantation is feasible for the preoperative assessment of patient-specific TVAR and provides the direct observation of the interaction between the prosthesis and the aortic valve. More advanced evaluation and validation are needed to perform a hemodynamic and structural mechanical simulation with multimodal imaging or in vitro model testing with 3D printing.


