Lots of interesting abstracts and cases were submitted for TCTAP 2023. Below are the accepted ones after a thorough review by our official reviewers. Don’t miss the opportunity to expand your knowledge and interact with authors as well as virtual participants by sharing your opinion in the comment section!
TCTAP A-072
Potential Genes and Pathways Related to the Foam Cell Formation Identified via Microarray Gene Expression Dataset Re-Analysis
By Jing Xu
Presenter
Jing Xu
Authors
Jing Xu1
Affiliation
Fuwai Hospital, China1
View Study Report
TCTAP A-072
Basic Science, Animal Models and Preclinical Studies
Potential Genes and Pathways Related to the Foam Cell Formation Identified via Microarray Gene Expression Dataset Re-Analysis
Jing Xu1
Fuwai Hospital, China1
Background
Atherosclerosis represents the leading abnormal condition of cardiovascular diseases. Foam cells play a crucial role in atherosclerotic lesion development, and macrophages and vascular smooth muscle cells (VSMCs) appear to be the majority of the cellular origins of foam cells formation.
Methods
Here we conducted an integrated microarray-based analysis based on GSE54666 and GSE68021 which contain samples of human macrophages and VSMCs incubated with oxidized low-density lipoproteins(ox-LDL). The differentially expressed genes (DEGs) in each dataset were investigated via the limma package in R. Geno ontology (GO) and pathway enrichment were performed via the ClueGO, CluePedia, and DAVID database. The convergent DEGs in the two cell types were obtained, and the protein interactions and transcriptional factors network were analyzed using STRING andTRRUST v2 database.
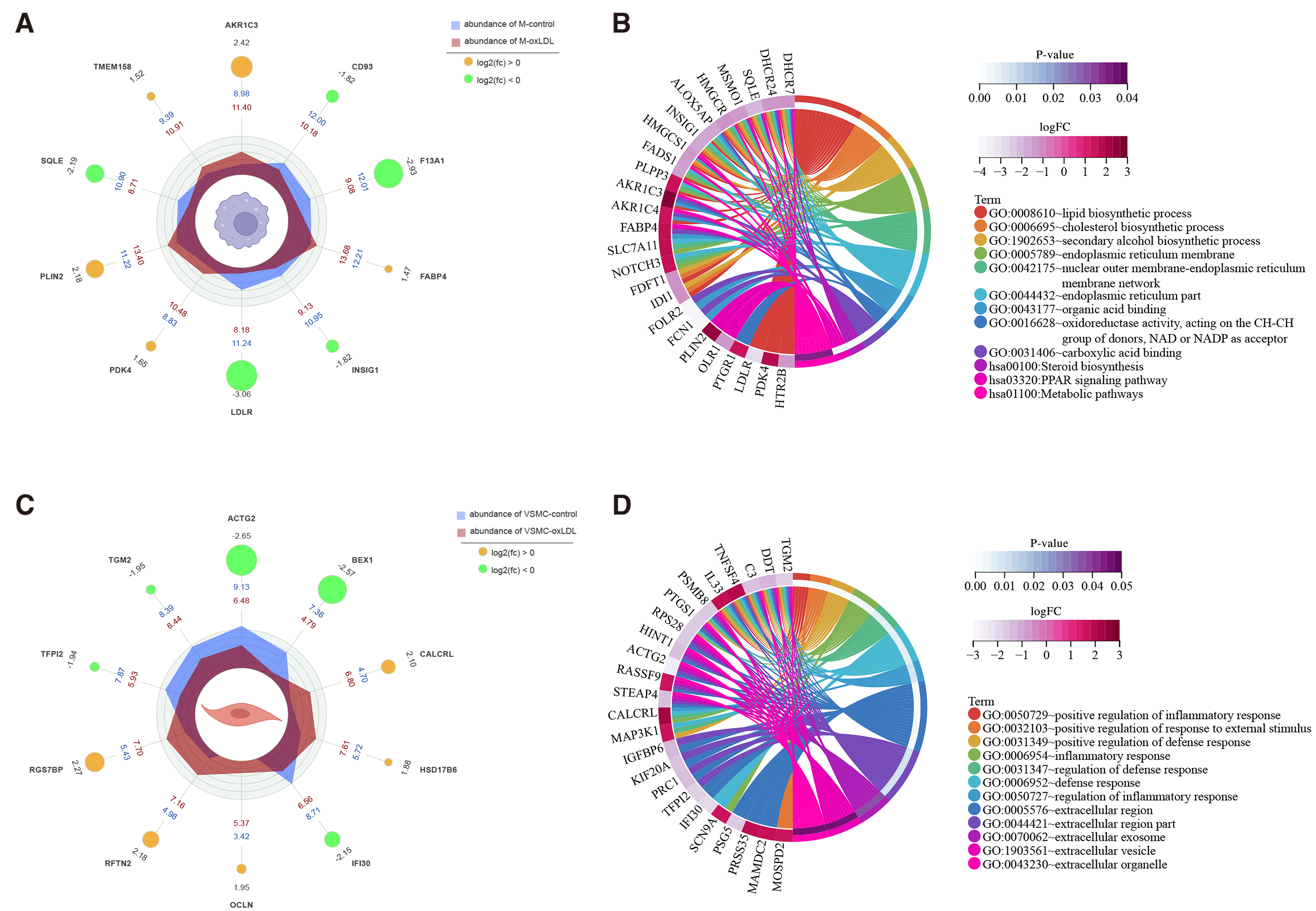

Results
For monocyte-derived macrophages, under the treatment of ox-LDL, a total of 504 DEGs were identified, and the top 5up-regulated DEGs were AKR1C3, PLIN2, PDK4, TMEM158,and FABP4, while the top 5 down-regulated DEGs were LDLR, F13A1,SQLE, INSIG1, CD93. For VSMCs, a total of 4400 DEGs were identified, and the top 5 up-regulated DEGs were RGS7BP, RFTN2, CALCRL,OCLN, HSD17B6, and the top 5 down-regulated DEGs were ACTG2,BEX1, IFI30, TGM2, TFPI2 in response to ox-LDLtreatment.
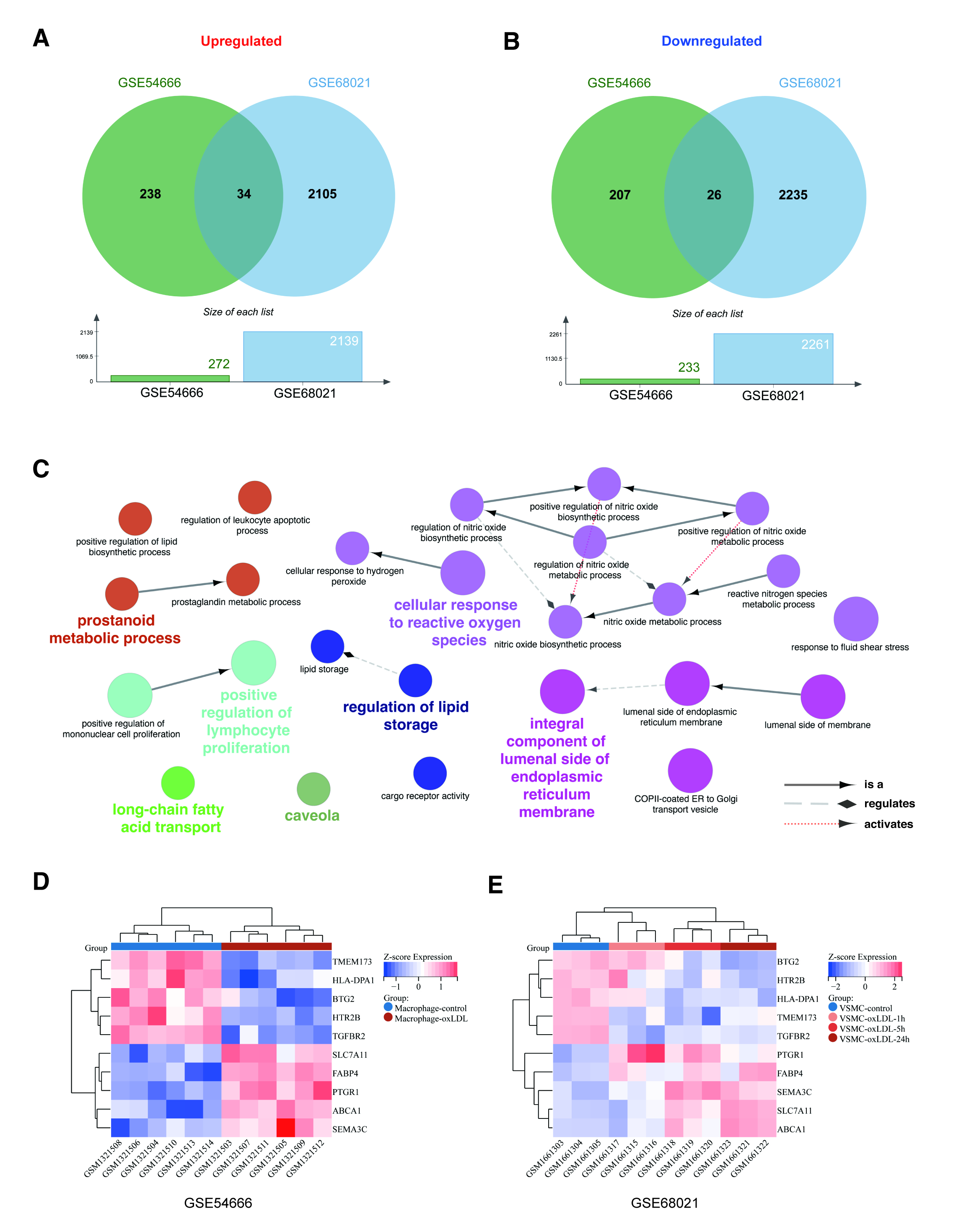

Conclusion
Our study has the merit of providing a comprehensive landscape of the transcriptional regulations in macrophages and VSMCs under the ox-LDL treatment from a bioinformatics respective, as well as suggesting several multi-cellular biomarkers and candidate pathways that might participate in the initial atherogenic events, which holds considerable promise to emerge capability to ascertain corresponding mechanisms and offer projections into developing new approaches for potential therapeutic modulation and cardiological practice.


