TCTAP A-072
Physiologic Lesion Assessment
Immediate Effect of Percutaneous Coronary Intervention by Bioresorbable Scaffold or Drug-eluting Stent on Superficial Wall Strain of Stenotic Coronary Arteries
Xinlei Wu1, Mick P.L. Renkens2, Laura Kerkmeijer2, Neil O’Leary3, Mattia Lunardi4, Jiayue Huang3, Daixin Ding3, Robbert De Winter5, Yoshinobu Onuma3, Patrick W. Serruys3, William Wijns3, Joanna Wykrzykowska2, Shengxian Tu6
The Second Affiliated Hospital of Wenzhou Medical University, China1, Amsterdam UMC, Netherlands2, University of Galway, Ireland3, National University of Ireland, Galway, Ireland4, Academic Medical Center, Netherlands5, Shanghai Jiao Tong University, China6
Background
In vivo quantification of the dynamic mechanical status of diseased coronary arteries may help for the prediction of cardiovascular outcomes. A new angiography-based method to quantify superficial wall dynamics during the cardiac cycle was developed and validated. The present study is aimed at investigating the immediate effect of stenting/scaffolding on superficial wall strain (SWS, dimensionless) before and after percutaneous coronary intervention (PCI).
Methods
Patients with de novo coronary artery lesions treated with bioresorbable scaffold (BRS, n=21) or drug-eluting stent (DES, n=21) were included in this study from the ABSORB Cohort B1 (n=14 patients) and the AIDA (n=23patients) trials. The SWS analyses were performed on the targeted vessels before and immediately after PCI. Measurements were quantified and peak SWS values were obtained within the stented segment and at 5 mm proximal and distal edges. The surface areas of hot spot-SWS were defined on the basis of the median SWS value on the entire vessel pre-PCI.
Results
A total of 82 SWS analyses were performed in 37 patients. At pre-PCI, the peak SWS on the ‘to be stented’ segment was significantly higher than the proximal and distal edges (0.79±0.37 versus 0.45±0.14 and 0.45±0.21; both p<0.001). The implantation of both devices led to a significant decrease (p<0.001) of the peak SWS by 0.44 (0.32, 0.51) [median (first, third quartiles)]. The surface area of high SWS decreased from 69.97 (28.02,111.92) mm
2 to 40.08 (17.10, 66.15) mm
2 (p=0.002). The peak SWS on the proximal and distal edges was not significantly affected. In vessel segments treated with BRS or DES, the peak SWS decreased to the same extent from 0.79 (0.56, 1.07) to 0.50 (0.35, 0.55) with BRS group (p<0.001) versus from 0.68 (0.54, 0.90) to 0.41 (0.30, 0.47) with DES group (p<0.001).
SWS_treated_DES_Post.aviSWS_treated_DES_Pre.avi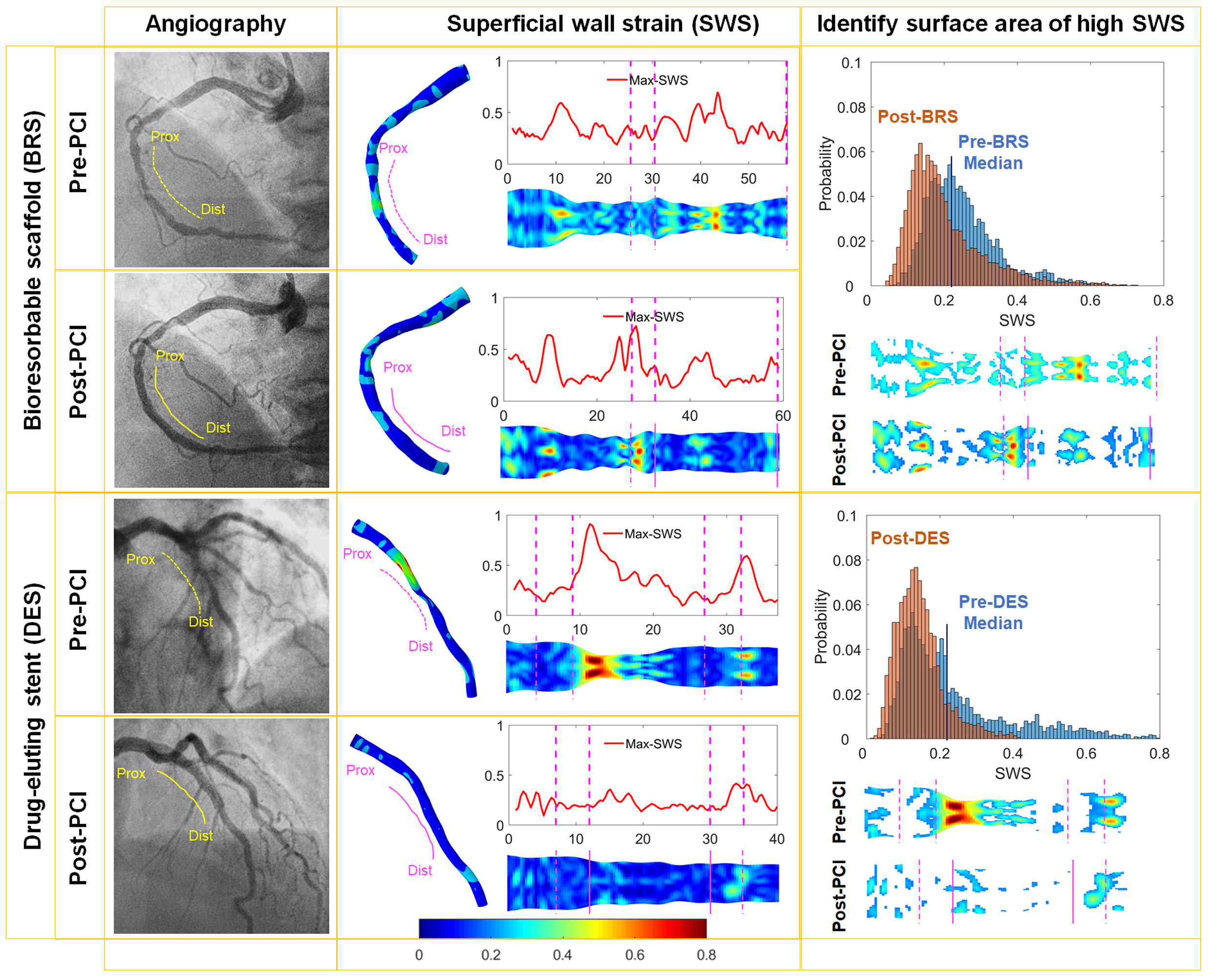
Conclusion
The quantitative assessment of SWS from angiography has the potential to provide valuable information about the mechanical status of the diseased arteries in vivo. Acutely, SWS at the treated segments was reduced similarly by metallic stents as by polymer-based scaffolds. Follow-up data will examine the predictive value of SWS on the long-term prognosis.



