Lots of interesting abstracts and cases were submitted for TCTAP 2021 Virtual. Below are accepted ones after thoroughly reviewed by our official reviewers. Don’t miss the opportunity to explore your knowledge and interact with authors as well as virtual participants by sharing your opinion!
TCTAP A-038
Presenter
Dae Young Hyun
Authors
Dae Young Hyun1, Myung-Ho Jeong2, Doo Sun Sim1, Dae Sung Park1, Munki Kim1, Xiongyi Han3, Kyung Hoon Cho1, Young Joon Hong1, Youngkeun Ahn1
Affiliation
Chonnam National University Hospital, Korea (Republic of)1, Gwangju Veterans Hospital, Korea (Republic of)2, Chonnam National University Hospital, China3
View Study Report
TCTAP A-038
Stents (Bare-metal, Drug-eluting)
Comparison of Mid- to Long-term Outcomes Between Polymer-free Everolimus-eluting Stents and Sirolimus-eluting Stents in a Porcine Model
Dae Young Hyun1, Myung-Ho Jeong2, Doo Sun Sim1, Dae Sung Park1, Munki Kim1, Xiongyi Han3, Kyung Hoon Cho1, Young Joon Hong1, Youngkeun Ahn1
Chonnam National University Hospital, Korea (Republic of)1, Gwangju Veterans Hospital, Korea (Republic of)2, Chonnam National University Hospital, China3
Background
Short-term data exist on the efficacy and safety of a polymer-free everolimus-eluting stent (EES) coated with a nitrogen-doped titanium dioxide (N-TiO2) film in a porcine coronary model. The long-term outcomes of this stent compared with those of a commercial non-polymer coronary stent have not yet been reported.
Methods
Sixty polymer-free EESs coated with an N-TiO2 film and polymer-free sirolimus-eluting stents (Coroflex ISAR®) were implanted in 30 pigs. Quantitative coronary analysis and optical coherence tomography (OCT) were performed immediately, 3 months, and 6 months after stenting. A histopathologic study was conducted at 1, 3, and 6 months after stenting.
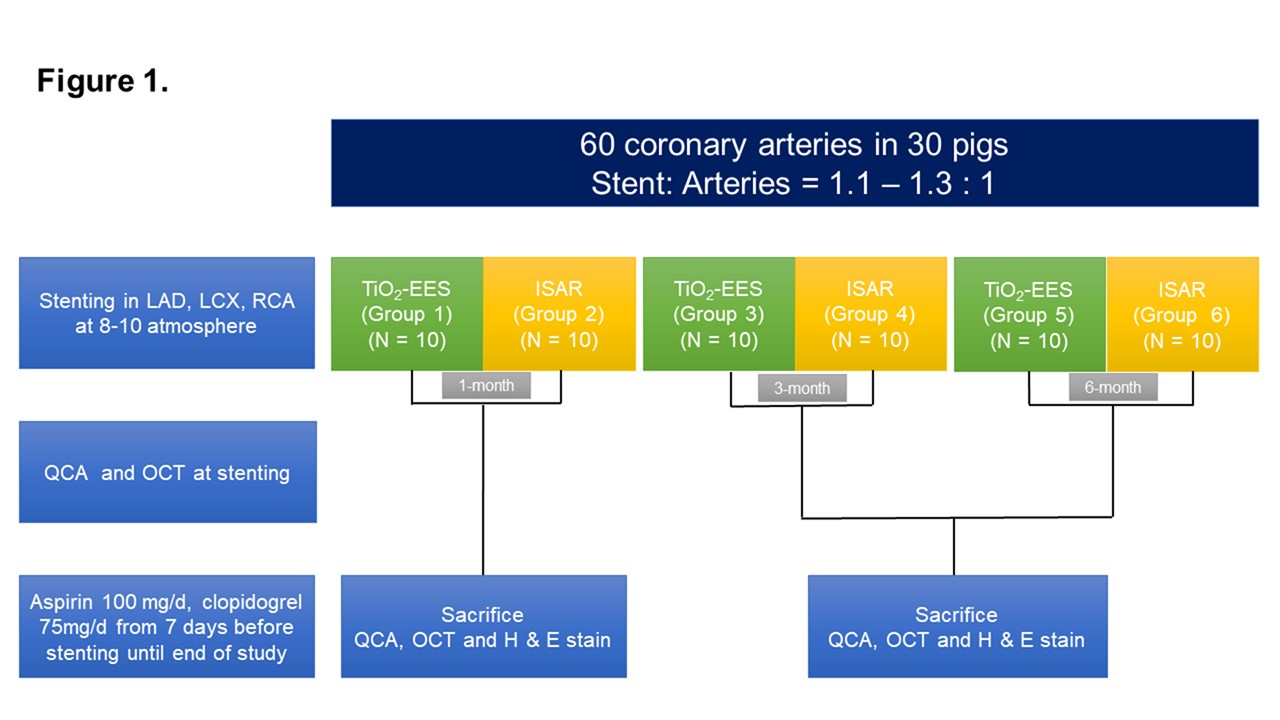

Results
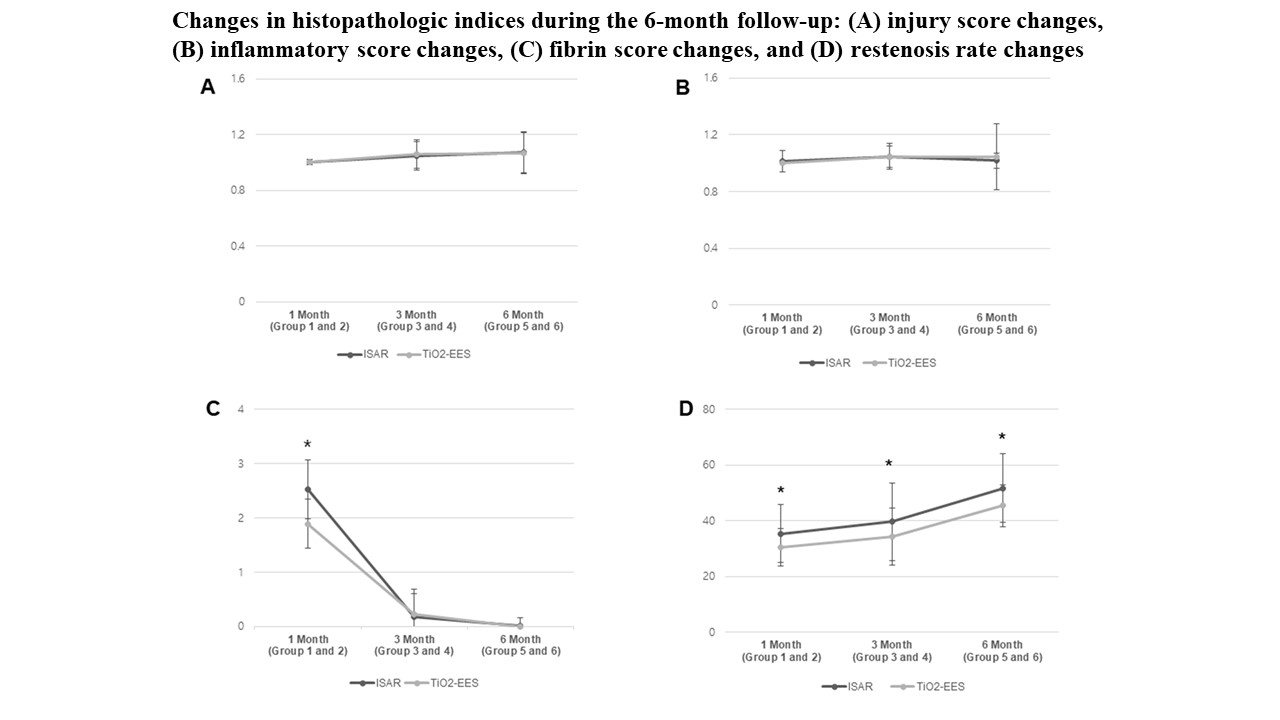
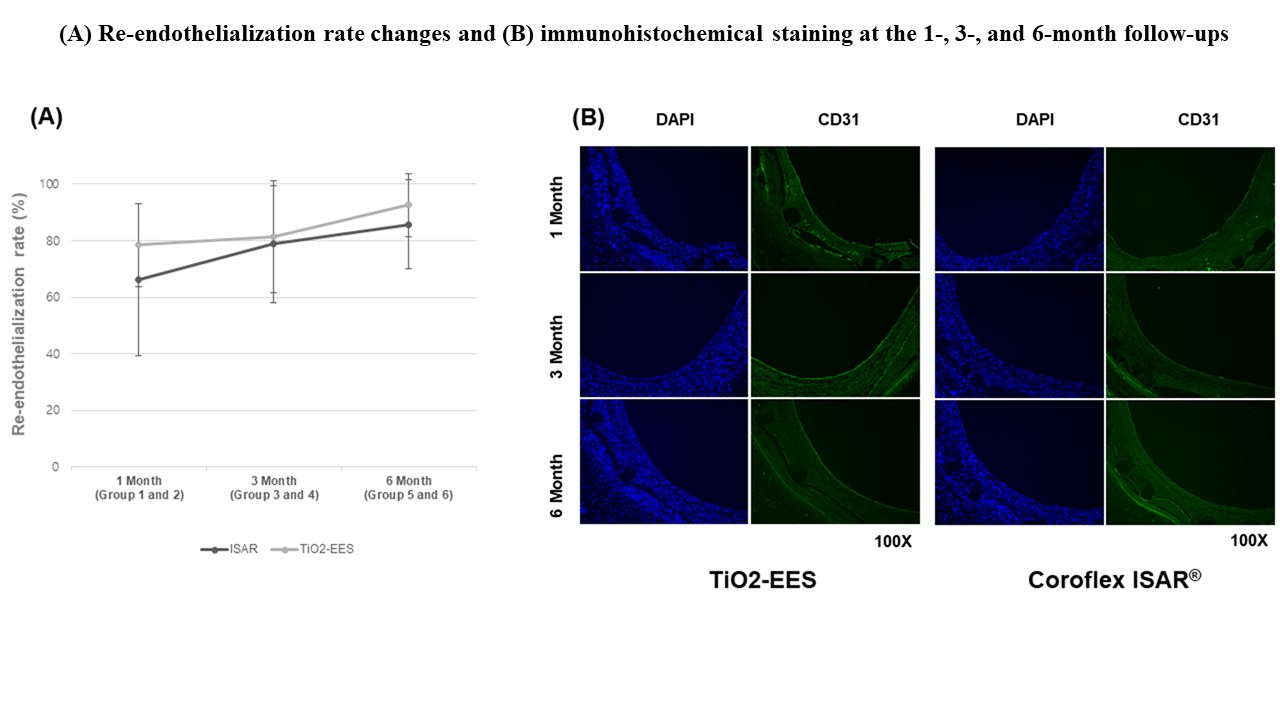
OCT at the 3-month follow-up showed a smaller neointimal area in the polymer-free EES group than in the Coroflex ISAR® group (22.5 ± 11.43% vs. 32.1 ± 12.27%, p< 0.001). Histopathology showed a lower fibrin score (1.9 ± 0.45 vs. 2.5 ± 0.54, p = 0.001) in the polymer-free EES group than in the Coroflex ISAR® group at 1 month. Re-endothelialization rates were not significantly different between the two groups. The polymer-free EES group had lower restenosis rates than the Coroflex ISAR® group over the entire follow-up period.
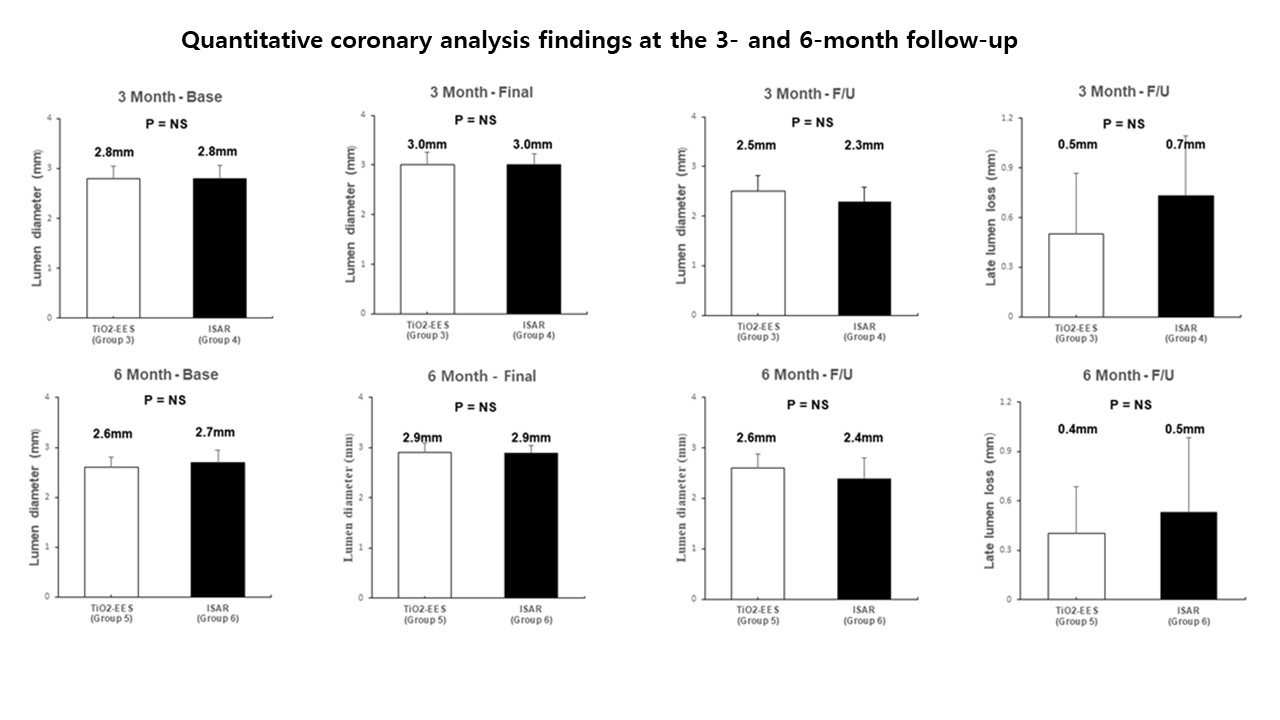
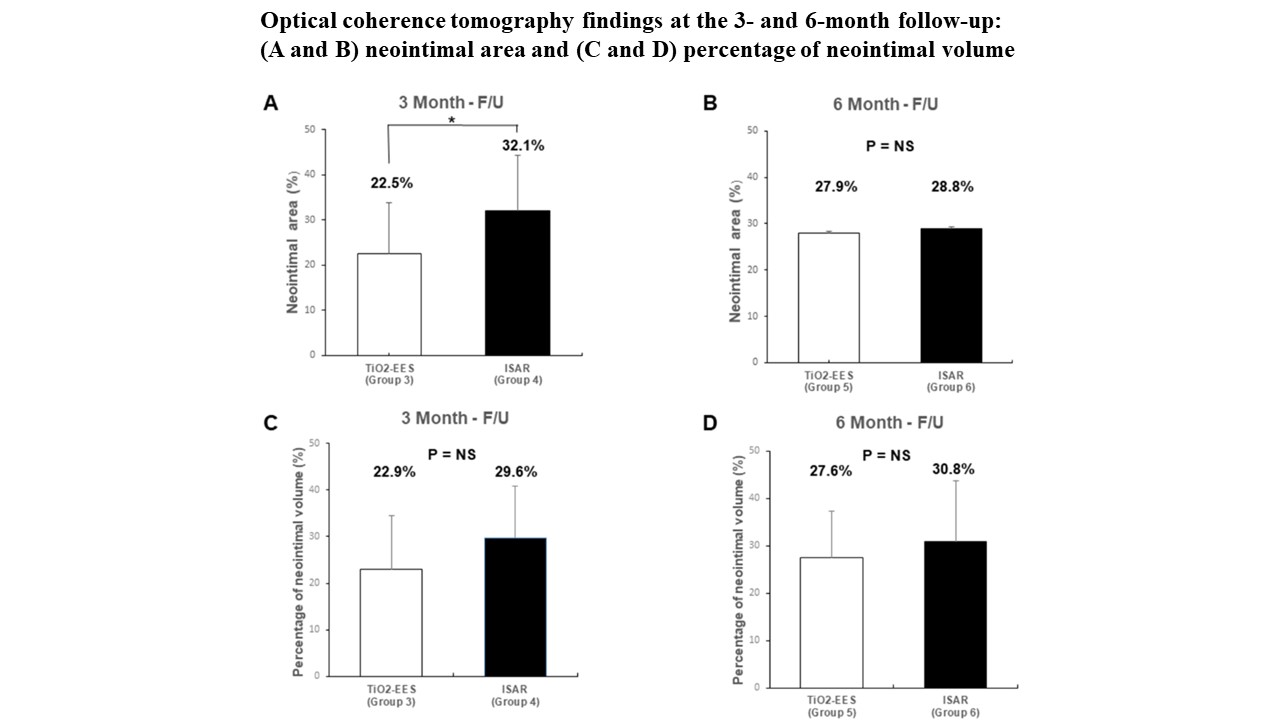
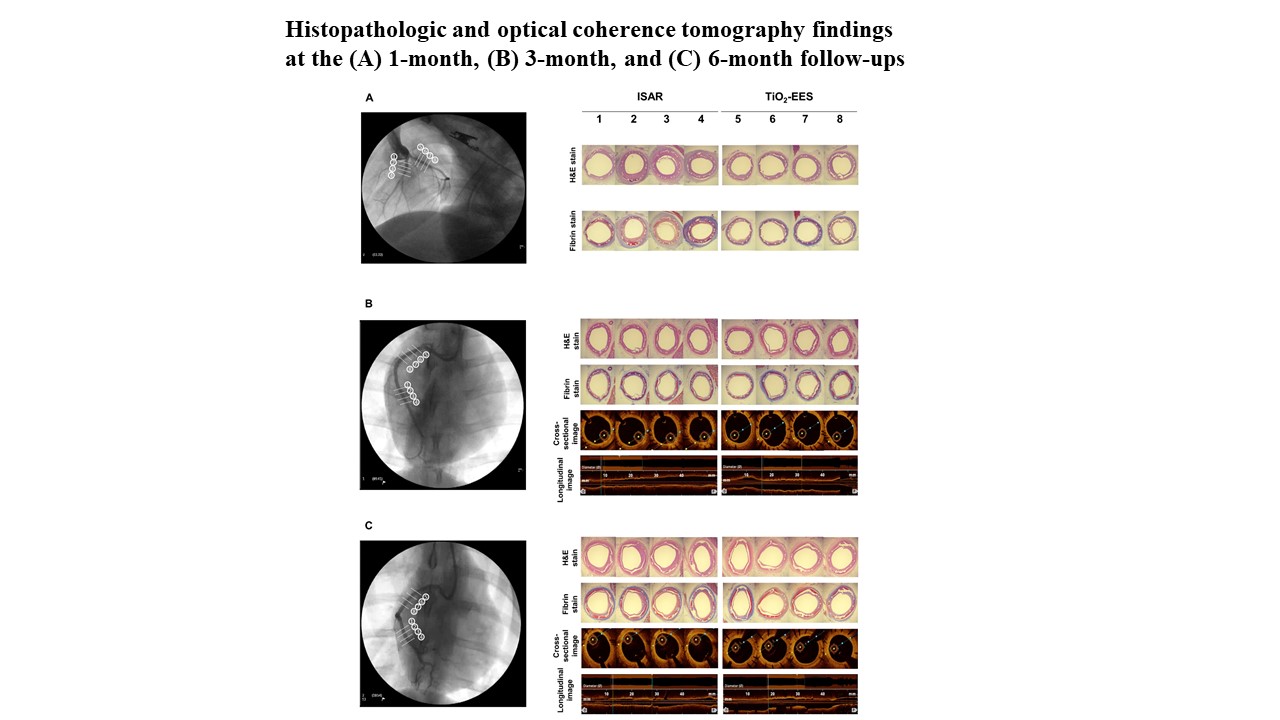





Conclusion
The novel polymer-free EES showed lower restenosis rates and higher biocompatibility than the Coroflex ISAR® on histopathology at the 6-month follow-up in a porcine model.


